Liquid filled in temperature sensors
Liquid fillers in temperature sensors use the principle of fluid expansion to measure temperature . If a liquid is trapped in a closed system and then heated, the molecules in those liquids put more pressure on the walls of the enclosed chamber. By measuring this pressure and / or allowing liquids to spread under constant pressure, we may infer the temperature of the fluid.
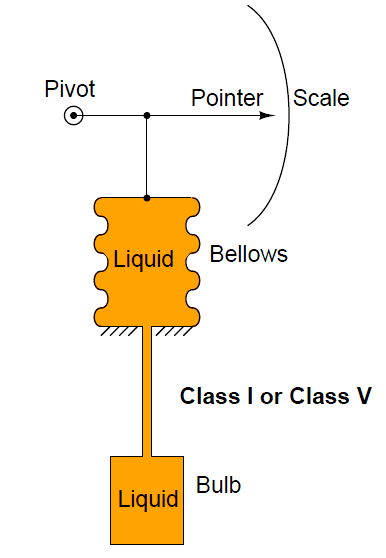
Four types of temperature sensor probes:
- Liquid Filled (Class IA, B
- (Vapor filled (Class II A, B, C, D
- (Gas Filled (Class III A, B
- (Mercury Filled (Class VA, B

Class I and Class V systems use a liquid filler (Class V is mercury).
Here, the volumetric expansion of the liquid has a marking mechanism to indicate the temperature:
Class III systems use gasoline filler instead of liquid. Here, changing the pressure with temperature (as described in the ideal gas law) allows us to feel the temperature of the probe:

In these systems, it is absolutely vital that the tube connecting the probe to the element indicates the minimum volume, so the expansion of the fluid is primarily due to a change in temperature in the probe, not a change in temperature along the tube. It is also important to understand that the volume of fluid in the rings (or Bourdon tube or diaphragm) is also subject to expansion and contraction due to temperature changes in this index. This means that the temperature indicator with the temperature change is somewhat different, which is not desirable, because we are going to do the temperature measuring device (exclusively) in the probe. There are various methods of compensation for this effect (for example, a bimetallic spring inside the indicator mechanism for automatic compensation of the signal by changing the ambient temperature), but it may be permanently compensated through a “zero” setting, provided The ambient temperature in the indicator does not change much.
A completely different class is the Class II filled probe system, which uses a volatile liquid / vapor combination to produce a temperature-dependent expansion of liquids:

Since liquid and vapor are in direct contact with each other, the pressure in the system will be exactly equal to the saturated vapor pressure at the vapor / liquid interface. This makes the Class II system sensitive to temperature only at the probe and nowhere else during system volume. Because of this phenomenon, a Class II-filled probe system does not need to compensate for temperature changes in the index. Class II systems have a special property, although they tend to change from Class IIA to Class IIB when the probe temperature exceeds the ambient temperature in the indicator.
Simply put, the liquid tends to follow the colder part of the Class II system while the steam tends to follow the warmer part. This problem occurs when the indicator and the identity of the probe exchange are felt as hotter or colder. The influx of liquid (or bottom) of the capillary tube as the system tries to reach a new equilibrium causes intermittent measurement errors. Class II probe systems designed to operate in either IIA or IIB modes are classified as IIC. A calibration problem for all systems with liquid-filled capillary tubes is compensation in temperature measurement due to hydrostatic pressure (or suction) caused by differences in the length of the measuring probe and the indicator. This indicates a “zero” change in calibration that may be offset by setting “zero” permanently during installation. Class III (gas-filled) and Class IIB (steam-filled) systems suffer from such a problem They do not cut because there is no fluid in the capillary tube to apply pressure due to the height.

