Compared to other gases, oxygen is highly paramagnetic. Oxygen paramagnetism can be thought of as the ability of an oxygen molecule to become a temporary magnet when placed in a magnetic field, similar to the magnetization of a piece of soft iron. The volumetric magnetic sensitivity of the flowing gas sample is felt in the detector / magnet set.
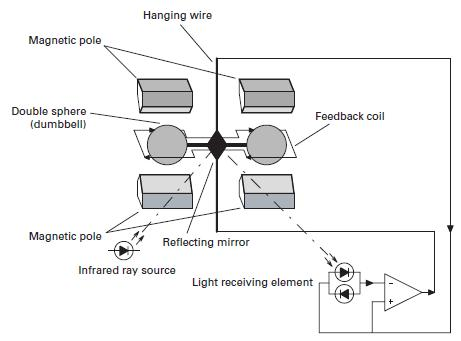
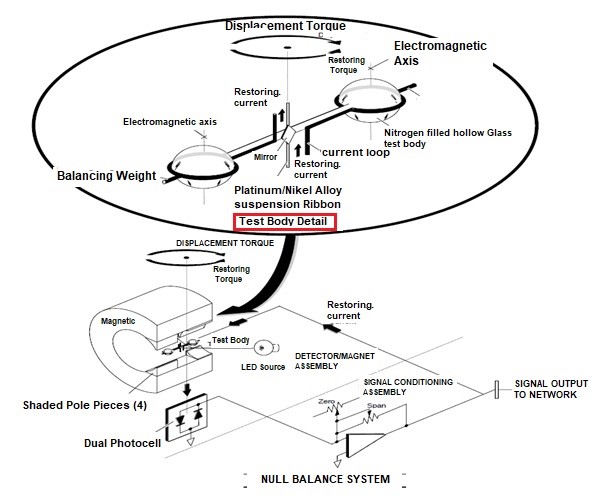
- As shown in the performance diagram of the figure, a hollow glass test body filled with dumbbell nitrogen is suspended on a platinum / nickel alloy strip in a non-uniform magnetic field.
- Due to the effect of magnetic buoyancy, the spheres of the test body are subjected to displacement forces, which leads to a displacement torque that is proportional to the volumetric magnetic sensitivity of the gas around the test body.
- The measurement is performed by a zero equilibrium system, where the displacement torque is equal to an opposite but opposite recovery torque.
- The restorative torque is caused by electromagnetic forces on the spheres created by the feedback current flowing through a titanium wire wrapped around the dumbbell.
- In fact, each sphere is wrapped with a circular loop of one rotation. The current required to return the test body to the zero position is directly proportional to the initial displacement torque and is a linear function of the volumetric magnetic sensitivity of the sample gas.
Performance diagram of O2 paramagnetic analyzer
What is an analyzer ?
- The recovery current is automatically maintained at the correct level by an electro-optical feedback system. A light beam from the source lamp is reflected from a square mirror attached to the test body and on a dual photocell.
- The output current of this compound is equal to the difference between the signals generated by the two half-photocells. This difference, which forms the error signal, is applied to the input of the amplifier circuit, which provides the recovery current. When the test body is in the zero position, both halves of the photocell light up equally. The error signal is zero. And the output of the amplifier remains constant.
- However, as soon as the test body starts to rotate, the amount of light becomes unequal and as a result an error signal is applied to the input of the amplifier circuit. The output amplifier signal is conducted through the current loop, thus generating the electromagnetic forces needed to return the test body to the zero position. In addition, the output of the amplifier is set to% O2 digital display and simultaneously sends 4-20 ma output.
- This analyzer is a dumbbell type paramagnetic oxygen analyzer. Because this analyzer is based on the fact that the magnetic sensitivity of oxygen gas is greater than that of existing gases, a stable measurement without the effect of existing gases is guaranteed. The detector does not have a heating part like a heater. Therefore, this analyzer is suitable for measuring the oxygen concentration in flammable gases. In addition, since reference gas is not required, current costs can be saved. The principle of measurement depends on the strong magnetic properties of oxygen molecules. Thus, measurement is almost magnetically weaker than oxygen under the influence of other molecules. Some of its features are as follows:
- Suitable for measuring oxygen in flammable gases.
- Small size and easy to handle.
- Can be used with a wide range of power supplies.
- The output is linear