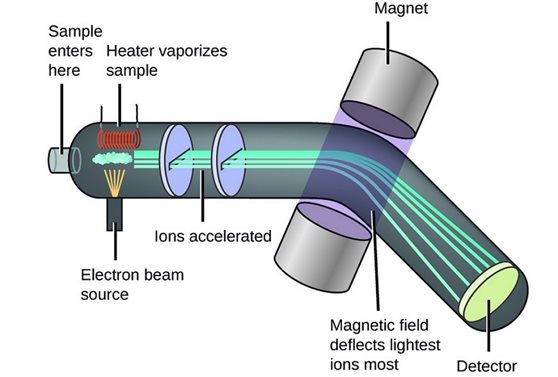
Mass spectrometry is one of the most practical analysis techniques in chemistry. This method is used for elemental analysis, surface analysis and for quantitative and qualitative purposes. The main components of the device include ion source, mass analyzer and Ionic Detector is. Various designs of this device are provided with various functions.
Principles of spectroscopy
Mass spectrometers perform three basic functions:
Molecules are bombarded by streams of energetic electrons, and some molecules are converted to their respective ions. Then the ions in an electric field Acceleration Are given.
Accelerated ions depending on their charge / mass ratio in one Magnetic field Or are electrically disconnected.
Ions with a specific charge / mass ratio are detected by a part of the device that is able to count ions when they collide with it. The output results are magnified and stabilized by the detector. The sign or role derived from stability is a mass spectrum, a graph of the number of particles detected as a function of the charge / mass ratio.
Mass Spectrometer
When we take a closer look at each operation, we see that the mass spectrometer is really more complex than what was described above. Prior to ion formation, the sample input system must find a way to direct a stream of molecules into the ionization chamber in which the ionization process takes place.
A sample input system is used to generate such a stream of molecules. Samples studied by mass spectrometry can be gaseous, liquid, or solid. In this method, devices must be used to vaporize a sufficient amount of the sample, then a stream of molecules is sent to the ionization chamber.
In the case of gases, matter itself exists in the vapor state. So, a simple input system can be used. The system is under vacuum, so that the ionization chamber is at a lower pressure than the sample inlet system.
The sample molecular orifice
goes to a larger warehouse from which the vapor molecules go to the ionization chamber. To ensure a uniform flow of molecules into the ionization chamber, steam passes through a small hole called a “molecular orifice” before entering. The same system is used for volatile liquids and solids. For low volatility materials, the system can be designed to be placed in an oven or oven to heat the sample.Pressure Generate more steam. Care must be taken not to damage the material with too much heat.
In the case of relatively non-volatile solids, a direct method can be used. The sample is placed at the tip of a rod and then enters the ionization chamber through a vacuum valve. The sample is located very close to the electron ionizing beam. The rod is then heated to produce heat from the sample to be expelled near the electron beam. Such a system can be used to study samples of molecules whose vapor pressure temperature The room is less than 9 – 10 mm Hg, used.
Ionization chamber
When a stream of sample molecules enters the ionization chamber, it is bombarded by a beam of energetic electrons. In this process, the molecules are converted to the corresponding ions and then accelerated in an electric field. In a beam ionization chamber, energetic electrons are emitted from a heated “thin wire”. This thin wire heats up to several thousand degrees Celsius. When operating under normal conditions, electrons have an energy equivalent to 70 microns-volts.
These energetic electrons collide with molecules that have entered the sample system and, by removing electrons from those molecules, ionize them and convert them into positive ions. A “repellent plate” that has a positive electrical potential directs new ions to a bunch of “accelerator plates”. A large potential difference (about 1 to 10 kV) is passed through these accelerator plates, which produces a beam of fast positive ions. These ions are led to a uniform beam by one or more “focusing gaps”.
Many sample molecules are not ionized at all. These molecules are continuously sucked in or Vacuum pumps Which are not connected to the ionization chamber, exit. Some of these molecules are converted to negative ions by electron capture. These negative ions are absorbed by the repellent plate. They may have a small fraction of ions formed more than once (losing more than one electron). These accelerate like monovalent positive ions.
Ionization potential
energy required to remove an electron from an atom or molecule is its ionization potential. Many organic compounds have an ionization potential of between 8 and 15 electron volts. But if the electron beam that hits the molecules does not have a potential of 50 to 70 electron volts, it will not be able to produce many ions. To create a mass spectrum, electrons with this amount of energy are used to ionize the sample.