- Zirconia oxygen analyzer is specifically used to measure the net concentration of oxygen in the flue gas. Measures the percentage of O2 by reading the voltage generated in a heated zirconium disk cell (750 ° C). When operating at the right temperature, the MV output of the cell is given by the equation.
E.M.F. (in mV) = KT log P1/P2 +C
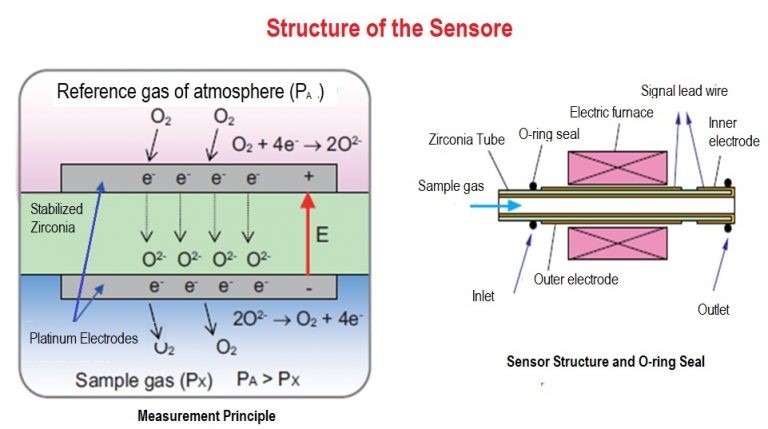
P2 is the partial pressure of oxygen in the flue gas on one side of the cell.
P1 is the partial pressure of oxygen in the reference gas on the other side of the cell. The air of the device is used as a reference gas.
T is the absolute temperature,
C fixed cell and
K is an arithmetic constant
- When the cell is at the right operating temperature and the oxygen concentration throughout the cell is unequal, O2 ions move from the partial pressure above O2 to the partial pressure below the cell. Since the output signal is proportional to the inverse partial pressure log of the sample O2, the output signal decreases with increasing O2 concentration. The output logarithm output voltage is approximately 53 MV / decade.
- A ceramic filter is provided at the tip of the O2 sensor probe to prevent the influx of carbon particles and dust into the sensor.
Zirconium oxide is a reliable and industry-proven measurement technology for the rapid and accurate analysis of oxygen in a wide range of applications, especially combustion processes.
Electronic converter
- The converter has a temperature controller, amplifier and power supply card.
- The electronic converter converts the MV output signal to a 4-20 MA signal that can be used as a signal.
- It also controls the temperature of the zirconia sensor at around (750 ° C) and gives the digital display% O2.
- The amplifier receives the MV signal generated by the sensor and produces a standard 4-20 Ma output signal.
Schematic diagram of O2 analyzer


