Temperature is a measure of the average molecular kinetic energy in a substance. This concept is easy for low-pressure gases, where gas molecules are randomly wrapped around it. The average kinetic energy of these gaseous molecules determines the temperature for that amount of gas. There is even a formula that expresses the relationship between average kinetic energy (Ek) and temperature (T) for a single gas (single atomic molecule)
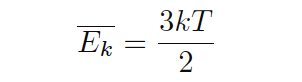
,Where
Ek = average kinetic energy of gaseous molecules (joules)
k = Boltzmann constant (10 – 23 1. 1.38 / Kelvin)
T = absolute gas temperature (Kelvin)
Thermal energy is a different concept
The amount of total kinetic energy for this random molecular motion. If the average kinetic energy is defined as 3kT / 2, then the total kinetic energy for all molecules in a monatomic gas must be the sum of the total number of molecules (N) in the gas sample

This may be expressed evenly in terms of the number of gasoline moles instead of the number of molecules (a considerably large number for each realistic sample)

,Where
Thermal = total thermal energy for a gas sample (joules)
n = amount of gas in the sample (mole)
R = ideal gas constant (8.315 joules per mole-Kelvin)
T = absolute gas temperature (Kelvin)
Heat is defined as the exchange of thermal energy from one sample to another through conduction (direct contact), convection (transfer through a moving fluid), or radiation (diffused energy). However, you will often find the terms heat and heat used interchangeably. For example, a single molecule of gas at a constant speed will have a certain temperature. Two or three molecules moving at the same speed have the same temperature, but together they have more thermal energy than each of them alone. Heat is the decrease or increase of thermal energy by energy transfer
If these gas molecules happen to move with slower molecules, the faster molecules will lose speed while the slower molecules will gain speed. Thus, higher temperature molecules are cooled while lower temperature molecules are heated: heat transfer. Temperature A smaller amount of heat energy or heat is detectable. In fact, when we need to measure heat energy or heat, we do this by measuring temperature and then inferring the desired variable according to the laws of thermodynamics
There are several methods for measuring temperature, from a simple glass lamp mercury thermometer to advanced infrared optical sensor systems. As with other measurements, there is no single technology that works best for all applications. Each method of measuring temperature has its own strengths and weaknesses. One of the responsibilities of a technician is to be aware of these positive and negative aspects in order to choose the best technology for application, and this knowledge is best achieved by knowing the operating principles of each technology
For information, you can read the article about temperature transmitters .
Temperature is the measure of how hot or cold something is. In particular, the measurement of the average kinetic energy of particles in an object, which is a type of motion-related energy. But how hot is it hot and how cold is it? Cold and hot terms are not very scientific terms. If we really want to determine how hot or cold something is, we have to use temperature. For example, how hot is molten iron? To answer this question, L, a physical scientist, measures the temperature of a liquid metal. Using temperature instead of words, such as hot or cold, reduces confusion.
Temperature depends on the kinetic energy of the particles
All matter is made up of particles – atoms or molecules – that are in constant motion. Because particles are moving, they have kinetic energy. The faster the particles move, the more kinetic energy they have. What does temperature have to do with kinetic energy? Well, as explained in this figure, the higher the kinetic energy of an object’s particles, the higher the body temperature.
 Low temperature
Low temperature
At lower temperatures, the particles move slower, have less kinetic energy, and produce less friction, so the heat is lower. Materials usually shrink, harden or freeze at low temperatures, and many materials change state. For example, at temperatures below zero degrees Celsius, liquid water freezes and turns to ice because there is no thermal energy.
 In many parts of the world, during the winter months, we experience colder temperatures in both poles of the earth. This is because the sun is farther away at these points / times.
In many parts of the world, during the winter months, we experience colder temperatures in both poles of the earth. This is because the sun is farther away at these points / times.
high temperature
At higher temperatures, the particles move faster and have higher kinetic energy, causing more friction and therefore more heat. Depending on the structure, materials may also begin to decompose, melt or expand.
 At hot temperatures, water changes state, turns into gas and evaporates. That’s why deserts are so common around the Earth’s equator – the hottest spot in the world.
At hot temperatures, water changes state, turns into gas and evaporates. That’s why deserts are so common around the Earth’s equator – the hottest spot in the world.
Temperature is an average measure. Particles of matter are constantly moving, but not all of them move at the same speed and in the same direction all the time. As we can see in this figure, the motion of the particles is random. Matter particles in an object move in different directions, and some particles move faster than others. As a result, some particles have more kinetic energy than others. So what determines the temperature of an object? The temperature of an object is the best approximation of the kinetic energy of the particles. When we measure the temperature of an object, we measure the average kinetic energy of the particles in the object.
The higher the temperature, the faster the molecules of matter move on average. Dyes disperse faster than hot water than cold water. This is due to the increased movement of molecules. Temperature is not related to the number of molecules involved. Under the given conditions, the temperature of 10 and 100 ml samples of boiling water is equal. This means that the average kinetic energy of the molecules is the same for two different amounts of water.
 The temperature of the tea in the cup and teapot is the same
The temperature of the tea in the cup and teapot is the same
In this picture, the tea in the teapot is higher than the glass, but the temperature of the tea in the glass is the same as the temperature of the tea in the teapot.
Temperature measurement
Because the molecules are so small, you have to use the indirect method to measure the kinetic energy of the molecules of a substance. When heat is added to a substance, the molecules move faster. This increase in motion causes a slight increase in the volume or amount of space occupied by most materials. There are devices that use the expansion of a substance to indirectly measure temperature. Such devices are called thermometers. There are several types of thermometers. Many thermometers are thin glass tubes filled with liquid. Mercury and alcohol are often used in thermometers because liquids remain in the wide temperature range. A change in temperature causes a small change in the volume of the liquid. However, this effect is magnified when the liquid expands in the very thin tube of the thermometer.

Some thermometers involve the use of bimetallic strips (temperature gauges). In such thermometers, strips made of two different metals are glued or glued together. Because metals expand at different speeds, the composite tape bends in a certain direction when heated. When cooled, it bends in the opposite direction. The figure below shows a bimetallic strip that is used as a thermostat. A thermostat is a device used to control heating and cooling systems.

Some thermometers, often used outside aquariums, contain liquid crystals that change color over time. As the temperature rises, more and more liquid crystal molecules collide with each other. This changes the structure of the crystals, which in turn affects their color. These thermometers are able to accurately determine the temperature between 65 degrees Fahrenheit and 85 degrees Fahrenheit.

A thermometer without a mark or rating will not be very useful to you. The thermometer is calibrated by marking two fixed points. The distance between these fixed points is divided into divisions called degrees. Degrees are used to indicate temperature. Three types of temperature scales are commonly used today: Celsius, Fahrenheit, and Kelvin. We are accustomed to expressing temperature in degrees Fahrenheit (F). Scientists often use degrees Celsius (C), but Kelvin (K) is the SI unit for temperature.
 Thermometers can measure temperature due to thermal expansion. Thermal expansion is the increase in volume of a substance due to an increase in temperature. As a substance heats up, its particles move faster. The particles themselves do not expand. They just spread to expand the whole material. Different materials expand for different values to change temperature. When you put the thermometer in a hot material, the liquid inside the thermometer expands and rises. You measure the temperature of a substance by measuring the expansion of a liquid in a thermometer.
Thermometers can measure temperature due to thermal expansion. Thermal expansion is the increase in volume of a substance due to an increase in temperature. As a substance heats up, its particles move faster. The particles themselves do not expand. They just spread to expand the whole material. Different materials expand for different values to change temperature. When you put the thermometer in a hot material, the liquid inside the thermometer expands and rises. You measure the temperature of a substance by measuring the expansion of a liquid in a thermometer.
Rose Calibration Company in Melbourne, Australia with over ten years of experience provides all calibration, maintenance, and repair services throughout Australia. If you live in Sydney, Melbourne, Adelaide, Perth, Geelong, and Brisbane, you can receive your quote in less than two hours by fill-up the form via the “Booking” link.